Dr. Paul Marik’s latest Cancer Care guide introduces a framework for simultaneously disrupting five metabolic systems in the cancer cell using repurposed drugs and nutraceuticals.

Every cancer treatment faces the same biological problem: cancer cells adapt. Block one energy source and they reroute to another. Cut off glucose and they lean on their mitochondria. Stress the mitochondria and they ramp up glycolysis. That metabolic flexibility is what allows tumors to survive under pressure, whether the pressure comes from chemotherapy, radiation, or repurposed drug protocols.
Repurposed drugs and metabolic therapies have brought new strategic options to this fight. Work by Dr. Paul Marik and others has shown that safe, affordable compounds can target cancer through mechanisms that conventional oncology largely ignores. But even metabolic therapies face a version of the same challenge: if the cancer cell has an open escape route, it will eventually find it.
The question is whether you can close them all at once. In his latest Cancer Care companion guide, The Metabolic Trap: Multi-Axis Metabolic Pressure in Cancer Therapy, Dr. Paul Marik lays out a coordinated five-axis framework designed to do exactly that: apply simultaneous pressure across five metabolic systems so the cancer cell has no viable fallback.
📘 This Guide Is Part of a Series
Dr. Marik’s cancer companion guides build on each other. For the best understanding, we recommend reading them in this order:
- The Approach to Repurposed Drugs for Cancer: Which drugs, which doses, and why.
- Cancer Resistance: When and how to cycle drugs to prevent treatment failure.
- The Metabolic Trap (you are here): Why targeting five metabolic axes at once can be decisive.
💡 Why We Made This Guide
Dr. Marik’s Cancer Care monograph laid the foundation for a metabolic approach to cancer treatment. Since then, a growing series of companion guides have translated that research into clinical action, covering everything from repurposed drug protocols to cancer stem cells to dietary interventions.
The companion guide on Cancer Resistance answered a critical question: when and how to cycle drugs to prevent treatment failure. This guide answers the next question: what if you could block every metabolic escape route at once?
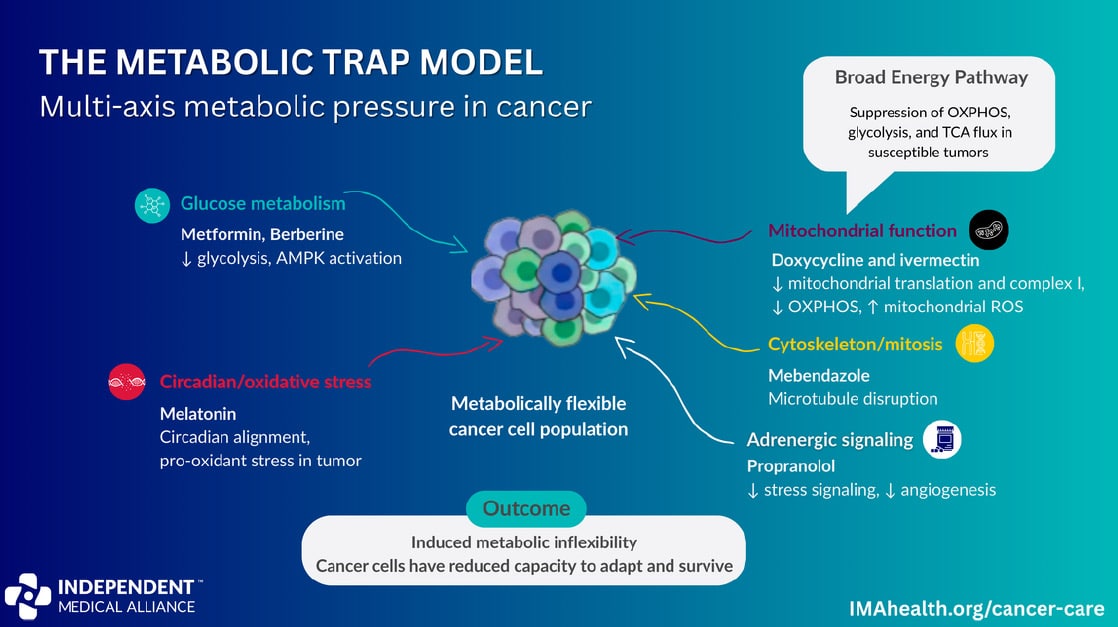
The concept is called multi-axis metabolic pressure. Rather than targeting one pathway and hoping the tumor doesn’t adapt, the framework applies coordinated pressure across five metabolic systems simultaneously. The goal is metabolic inflexibility: a state where the cancer cell has no remaining fuel source, no compensatory pathway, and no way to outlast the protocol.
“With these five used in combination, you completely whack-a-mole the cancer cell. It has no way of getting energy and surviving.” — Dr. Paul Marik
For the full video discussion of both this guide and the companion Cancer Resistance guide, watch the recent webinar: Drug Cycling in Cancer Care: What It Is and Why It’s Critical.
🔬 The Five-Axis Framework
Cancer cells normally adapt to metabolic pressure by switching fuels: glucose, glutamine, fatty acids. When you block one source, the cell pivots to another. That flexibility is what makes single-pathway treatments vulnerable over time.
The Metabolic Trap targets five systems at once:
- Glucose metabolism: Metformin and berberine activate the AMPK energy sensor, suppress mTOR signaling, and reduce the tumor’s access to its primary fuel source.
- Mitochondrial function: Doxycycline inhibits mitochondrial translation, impairing the energy production that cancer stem cells depend on for persistence and relapse.
- Cytoskeleton/mitosis: Mebendazole disrupts tubulin polymerization and mitotic spindle function, placing pressure on the machinery required for cell division.
- Adrenergic signaling: Propranolol blocks beta-adrenergic receptors on tumor cells, reducing stress-mediated invasion, angiogenesis, and immune suppression.
- Circadian/redox regulation: Melatonin restores circadian alignment and increases oxidative stress within the tumor, making it harder for cancer cells to exploit redox adaptation as a survival mechanism.
When multiple axes are suppressed simultaneously, tumor cells lose their ability to adapt. The result is energetic collapse, impaired proliferation, and increased susceptibility to cell death. The combination doesn’t work because each drug is individually powerful; it works because together they eliminate the cancer cell’s options.
“If you have constant pressure on that pathway, just by evolutionary biology, the cells will adapt and mutate, so they become resistant.” — Dr. Paul Marik
That quote describes the problem. The five-axis framework is the solution.
🧩 Blocking the Escape Routes
The guide makes a critical distinction that connects directly to the Cancer Resistance cycling strategy.
Single-pathway drugs like doxycycline and mebendazole are vulnerable to resistance under sustained pressure. They need to be cycled, alternating monthly in an eight-week rotation so the tumor can never fully adapt to either one. Multi-pathway agents like metformin, berberine, propranolol, and melatonin hit multiple targets at once, making resistance far harder to develop. They run continuously through both phases of the cycle.
Ivermectin occupies a unique role in the framework. The guide describes it as a bridge drug rather than a sixth isolated axis. Preclinical research links it to effects across multiple systems: Wnt signaling, AKT-mTOR, drug efflux pumps, and mitochondrial stress. It reinforces several parts of the trap simultaneously, connecting the glucose axis to the mitochondrial and stress-response axes.
The logic is straightforward. If glucose pressure increases, tumors lean on mitochondria. If mitochondrial pressure increases, they ramp up glycolysis. If stress hormones support invasion, cells survive despite metabolic pressure. A coordinated combination blocks several of these escape routes at once, leaving the cancer cell with fewer places to go. That is the biochemical logic of the trap.
All of the agents in the framework are inexpensive, widely available, and carry well-established safety profiles. The guide is designed to be used alongside a clinician who can adapt the strategy based on individual response.
📚 More Cancer Care Resources
- New Guide: The Metabolic Trap: Multi-Axis Metabolic Pressure in Cancer Therapy
- Companion Guide: Cancer Resistance and Interventions to Mitigate Resistance
- Monograph: Cancer Care
- Hub: IMA Cancer Resource Hub
- Webinar: Drug Cycling in Cancer Care: What It Is and Why It’s Critical
- Guide: The Approach to Repurposed Drugs for Cancer
- Guide: Targeting Cancer Stem Cells
- Guide: Dietary Interventions in Cancer