Treatment resistance is one of the biggest unsolved problems in cancer care. Dr. Paul Marik’s latest guide explains why it happens and introduces a practical cycling strategy designed to prevent it.

Every cancer patient eventually faces the same fear: what happens when the treatment stops working?
It’s a question that haunts oncology. Conventional therapies like chemotherapy and radiation frequently lose effectiveness over time. Tumors shrink, patients improve, and then the disease roars back. The medical establishment has known about treatment resistance for decades, but patients are rarely told why it happens or given a strategy to prevent it. They’re simply moved to the next line of therapy and told to hope for the best.
Repurposed drugs and metabolic therapies have brought new hope to that fight. Work by Dr. Paul Marik and others has shown that safe, affordable compounds can target cancer through mechanisms that conventional oncology largely ignores.
Treatment resistance, however, doesn’t discriminate. It’s a biological reality that affects chemotherapy, radiation, targeted drugs, and metabolic therapies alike. The difference is that the metabolic framework now has a strategy to address it. In his latest Cancer Care companion guide, Cancer Resistance and Interventions to Mitigate Resistance, Dr. Paul Marik explains why cancer cells adapt under treatment pressure and lays out a practical, evidence-based cycling strategy to stay ahead of them.
📘 This Guide Is Part of a Series
Dr. Marik’s cancer companion guides build on each other. For the best understanding, we recommend reading them in this order:
- The Approach to Repurposed Drugs for Cancer: Which drugs, which doses, and why.
- Cancer Resistance (you are here): When and how to cycle drugs to prevent treatment failure.
- The Metabolic Trap: Why targeting five metabolic axes at once can be decisive. (Full post coming soon.)
💡 Why We Made This Guide
Dr. Marik’s Cancer Care monograph laid the foundation for a metabolic approach to cancer treatment. Since then, a growing series of companion guides have translated that research into clinical action, covering everything from repurposed drug protocols to cancer stem cells to dietary interventions.
This guide tackles a question the others hadn’t addressed: what happens when a metabolic protocol stops working?
The question that sparked this guide came from the front lines. A physician reached out to Dr. Marik with a troubling pattern: his cancer patients were responding well to repurposed drug therapy for up to two years, and then their tumors would spiral out of control. Nothing he adjusted could restore the response. That clinical reality sent Dr. Marik on a research path that led to a surprising discovery.
The answer, it turns out, has been hiding in plain sight. The Care Oncology Clinic (COC) protocol, a well-known four-drug metabolic regimen, was designed with a specific cycling pattern built in. But the published literature never highlighted why that cycling mattered. Dr. Marik dug into the evolutionary biology behind it and built a comprehensive framework, backed by 32 peer-reviewed references, that any clinician can apply.
“If you have constant pressure on that pathway, just by evolutionary biology, the cells will adapt and mutate, so they become resistant.” — Dr. Paul Marik, IMA Chief Scientific Officer
For the full video discussion of the guide, including a companion guide on a related concept called The Metabolic Trap, watch the recent webinar: Drug Cycling in Cancer Care.
🔬 How Tumors Outsmart Therapy
Cancer cells are not passive. Under chronic metabolic pressure from repurposed drugs and nutraceuticals, they don’t simply die or hold steady. They adapt.
Think of it this way: if you apply the same stress to a tumor for months on end without varying your approach, you’re giving it time and stability to find workarounds. The cells that survive are the ones best equipped to rewire their metabolism, enter dormant states, or exploit alternative fuel sources. Over time, those survivors take over.
The guide identifies five key biological drivers behind this process:
- Metabolic switching: Cancer cells toggle between glucose-based and oxygen-based energy production, finding whichever fuel source your protocol isn’t blocking.
- Cancer stem cell enrichment: A small, resilient subpopulation of stem-like cells survives therapy and seeds regrowth.
- Stress signaling and epigenetic rewiring: Cells activate backup survival programs that become permanently baked in over time.
- Microenvironmental buffering: The tumor’s surrounding tissue, including immune cells and connective tissue, adapts to protect it.
- Genetic hardening: Repeated stress selects for clones with mutations that lock in resistance permanently.
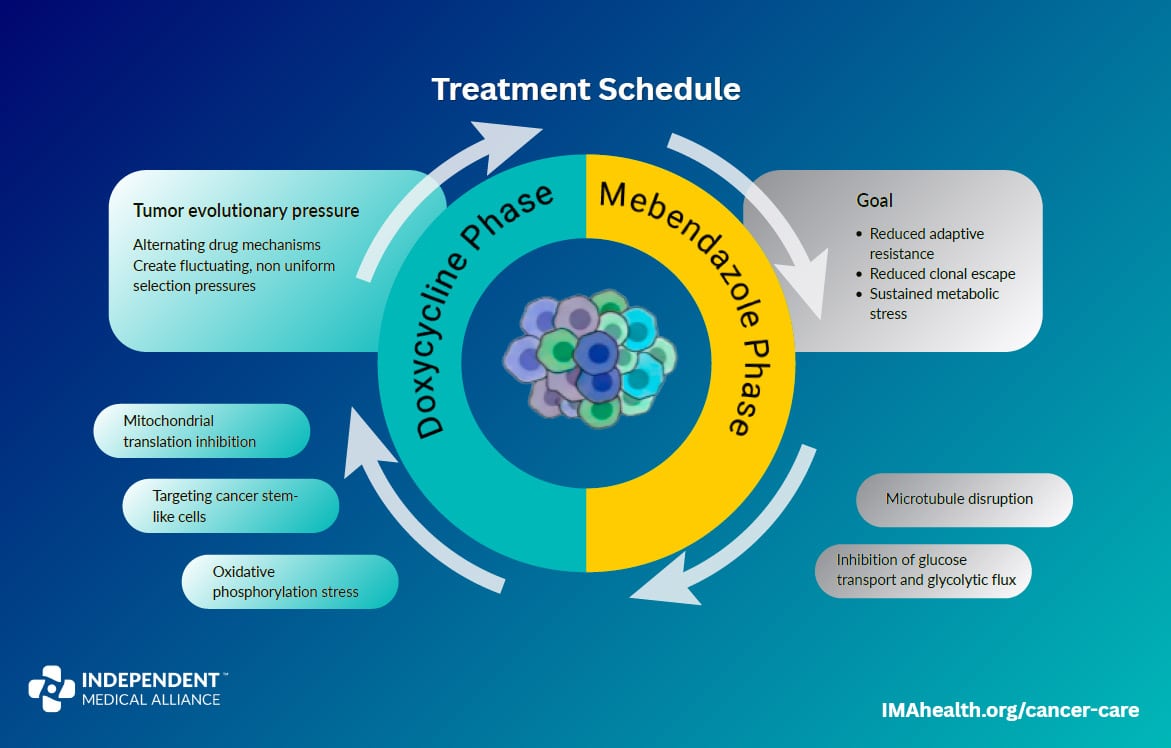
The critical takeaway: resistance isn’t a sign that metabolic therapy doesn’t work. It’s a sign that the protocol needs to evolve along with the tumor.
🎯 The 8-Week Cycling Strategy: What to Rotate and What to Keep Running
Not all drugs in a metabolic protocol carry the same resistance risk. Some target a single cellular pathway, which means the tumor can eventually adapt around the blockade if the pressure never changes. Others act across multiple pathways simultaneously, making it far harder for any one resistant clone to take over. The guide draws a clear line between the two.
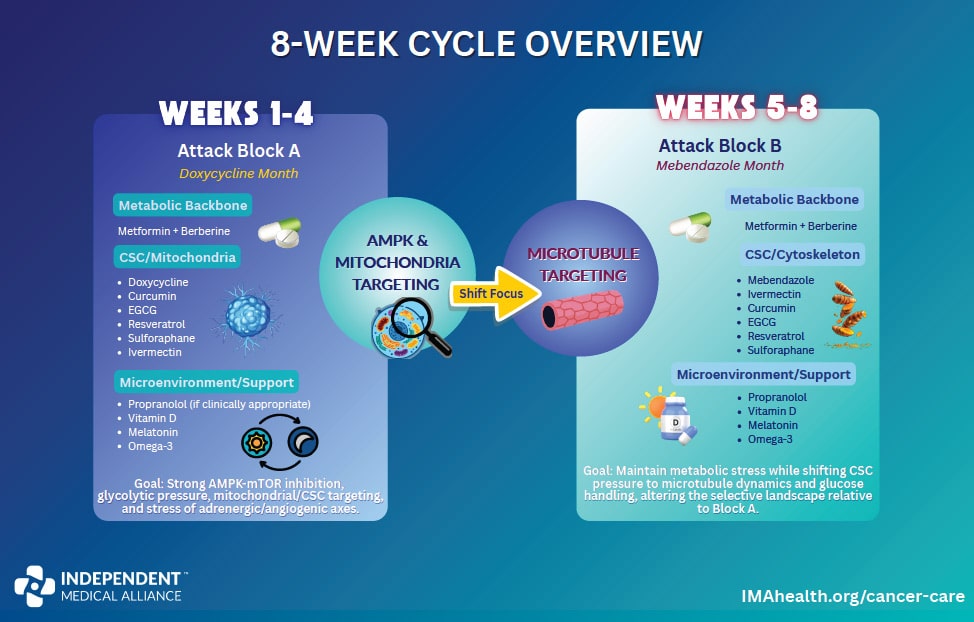
The centerpiece of the strategy is an eight-week adaptive cycle that rotates the two single-pathway agents while keeping everything else running continuously.
Rotate Monthly: Doxycycline and Mebendazole
These two drugs each target one dominant cellular system. Doxycycline inhibits mitochondrial translation, cutting into the cancer cell’s energy production. Mebendazole disrupts microtubules, blocking the cell’s ability to divide and maintain its internal structure. Used continuously, either one gives the tumor time to adapt around a single blockade. Rotated monthly, they exploit a phenomenon called collateral sensitivity: cells that adapt to resist mitochondrial stress become more vulnerable to microtubule disruption, and vice versa.
Weeks 1–4 (Block A): Doxycycline runs alongside the continuous backbone.
Weeks 5–8 (Block B): Mebendazole swaps in; doxycycline comes off.
This approach draws from the Care Oncology Clinic protocol, which was designed with this rotation built in, and from established principles in adaptive therapy and tumor evolutionary biology.
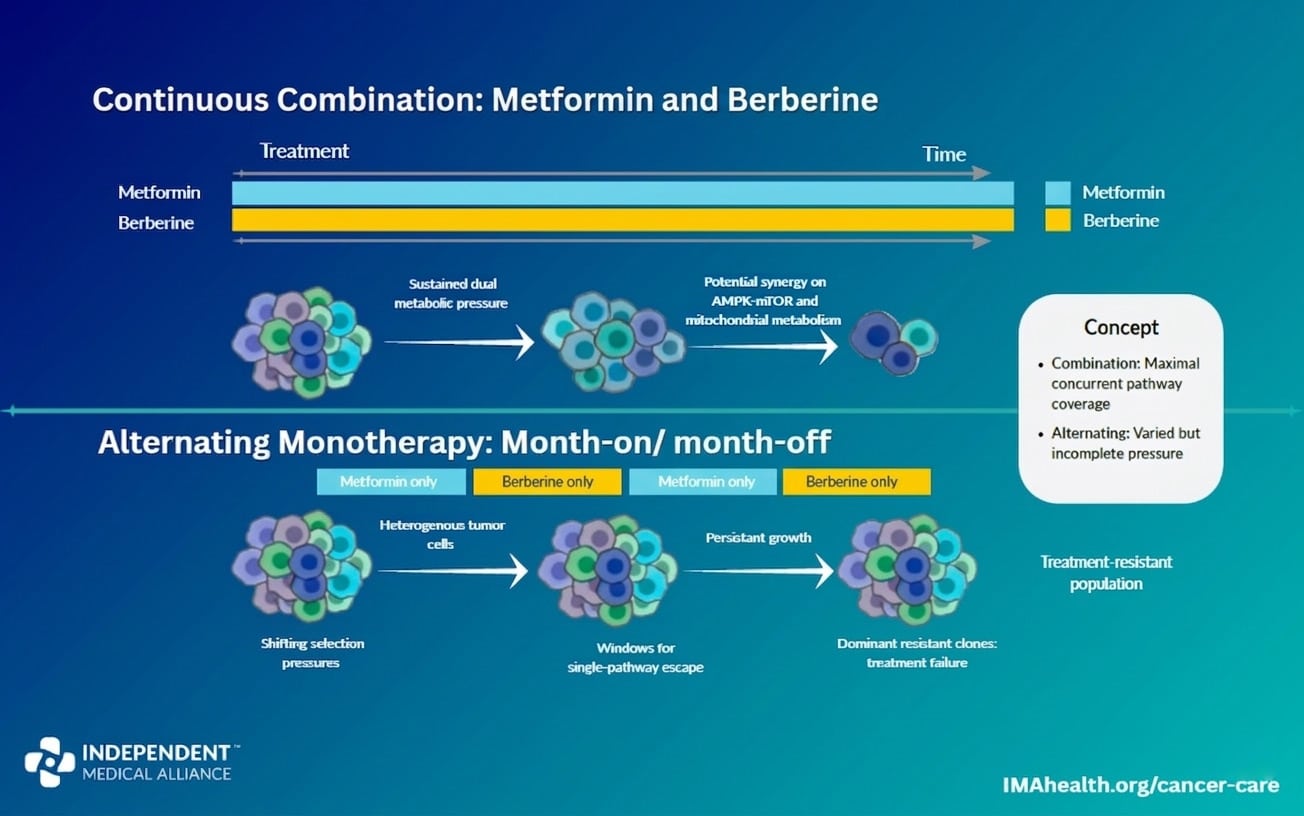
Keep Continuous: Metformin and Berberine
Both activate the AMPK energy sensor, but their systemic effects are complementary rather than redundant. Metformin works primarily on liver metabolism and insulin signaling; berberine exerts stronger direct cellular effects and modulates the gut microbiome. Together they produce stronger metabolic restriction than either alone. Because resistance to these agents arises from broad metabolic adaptation rather than single-pathway mutations, continuous dual pressure is more effective than cycling between them.
Keep Continuous: Ivermectin
Unlike single-pathway drugs, ivermectin acts across multiple targets simultaneously, including Wnt signaling, PI3K/AKT/mTOR, drug efflux pumps, and mitochondrial stress. That multitargeted activity makes it difficult for tumors to develop a dominant resistance pathway. It also functions as a sensitizer, making other therapies more effective.
Keep Continuous: The Core Phytochemicals
Curcumin, EGCG, sulforaphane, and resveratrol are broad, multitarget signaling modulators. Their role is persistent suppression of inflammatory, metabolic, and pro-survival signaling rather than direct cytotoxicity. There is no clinical evidence that rotating them prevents resistance, though some clinicians alternate for practical or GI tolerance reasons.
The Continuous Backbone (Runs Through Both Blocks)
In addition to metformin, berberine, ivermectin, and the phytochemicals above, the following agents run continuously throughout both four-week blocks: propranolol (if clinically appropriate), vitamin D, melatonin, and omega-3 fatty acids.
🐟 Omega-3 Fatty Acids: A Resistance-Reducing Role
One finding from the guide worth highlighting: omega-3 fatty acids (EPA and DHA) may play a specific role in reducing treatment resistance. They incorporate into tumor cell membranes, increasing membrane fluidity and improving drug penetration into the cell. They also suppress P-glycoprotein, the efflux pump that cancer cells use to expel drugs before they can do damage.
Perhaps most importantly, omega-3-enriched membranes become vulnerable to a form of oxidative damage that can trigger cell death through a process called ferroptosis. The omega-3s don’t kill the cancer directly; they make the cancer cell’s membranes more fragile so that other therapies hit harder. The guide recommends 2–4 g/day of combined EPA + DHA.
🧩 The Bigger Picture
Cancer Resistance answers the question of when and how to sequence drugs to prevent treatment failure. Its companion guide, The Metabolic Trap, answers the question of why targeting five metabolic axes at once can be decisive. Together they form a unified strategy: apply multi-axis metabolic pressure so the cancer cell has no viable energy source, and cycle the single-pathway agents so it can’t adapt its way out.
Both guides are free, both are backed by peer-reviewed research, and both are designed to be used alongside a clinician. For the full video discussion, watch the recent webinar: Drug Cycling in Cancer Care: What It Is and Why It’s Critical.
📚 More Cancer Care Resources
- New Guide: The Metabolic Trap
- Monograph: Cancer Care
- Hub: IMA Cancer Resource Hub
- Webinar: Drug Cycling in Cancer Care: What It Is and Why It’s Critical
- Guide: The Approach to Repurposed Drugs for Cancer
- Guide: Targeting Cancer Stem Cells
- Guide: Dietary Interventions in Cancer
- Guide: Preoperative Repurposed Drugs